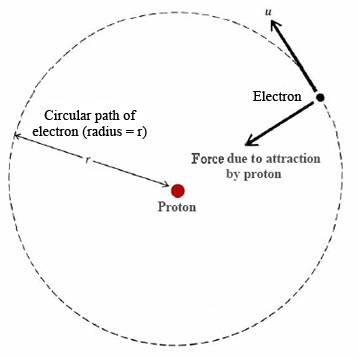
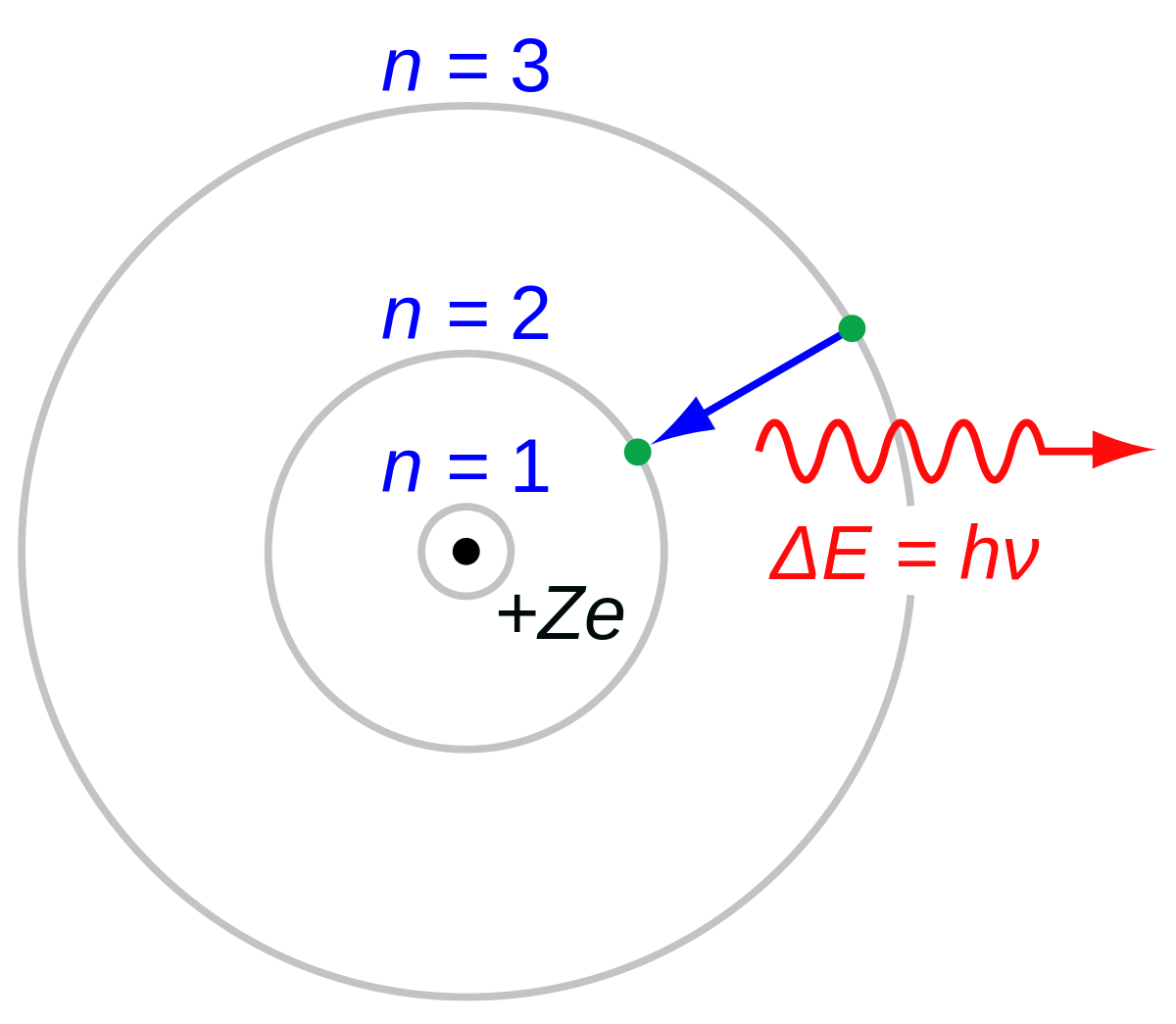
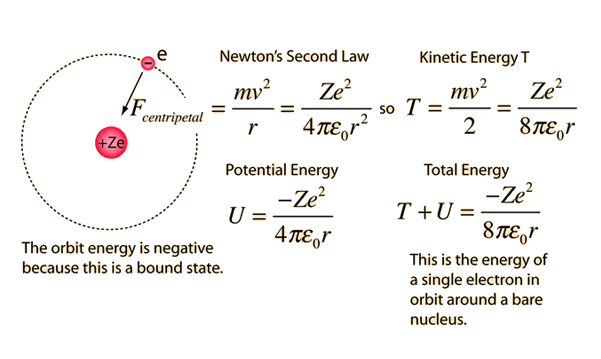
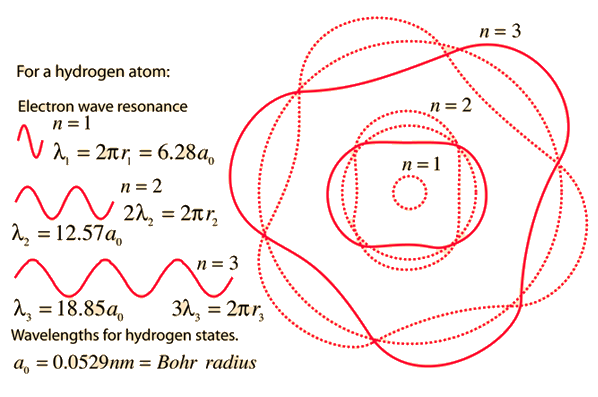
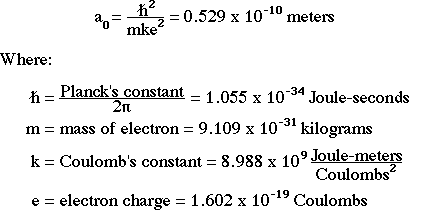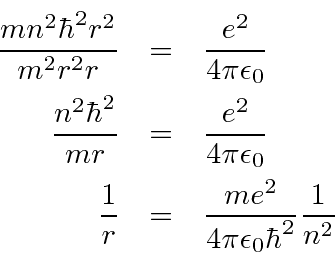Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

Show all steps of the derivation: Show that the most probable radius for an electron described by the function R_10 is the Bohr radius, a_0. | Homework.Study.com

Derivation of Atomic Radius Of Bohr Orbit ForJEE Mains /Advanced & NEET /AIIMS BySukhdev Patsariya | - YouTube
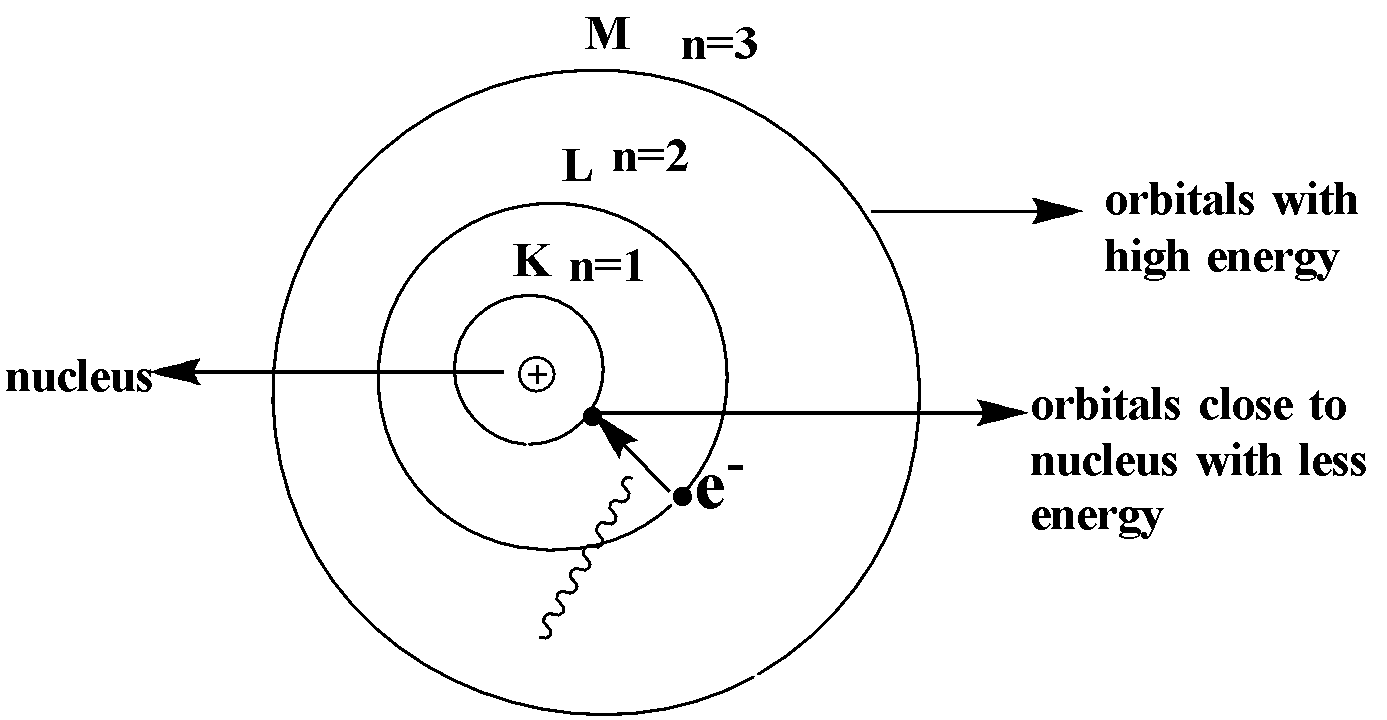
In Bohr's atomic model, radius and energy in orbit (n) is related as:A. $r \\propto {n^2}$, $E \\propto \\dfrac{1}{{{n^2}}}$B. $r \\propto \\dfrac{1}{{{n^2}}}$, $E \\propto {n^2}$C. $r \\propto \\dfrac{1}{n}$, $E \\propto n$D. $E \\
What is the most possible radius (in PM) for an electron in the first orbit of a hydrogen atom? - Quora

1. The first Bohr's radius (for electron in hydrogen atom in the ground state): 2. The ground energy level in hydrogen atom: - ppt video online download

If the radius of Bohr's second orbit of hydrogen atom is alpha then the radius of the 4^th Bohr orbit in atom with Z = 3, is