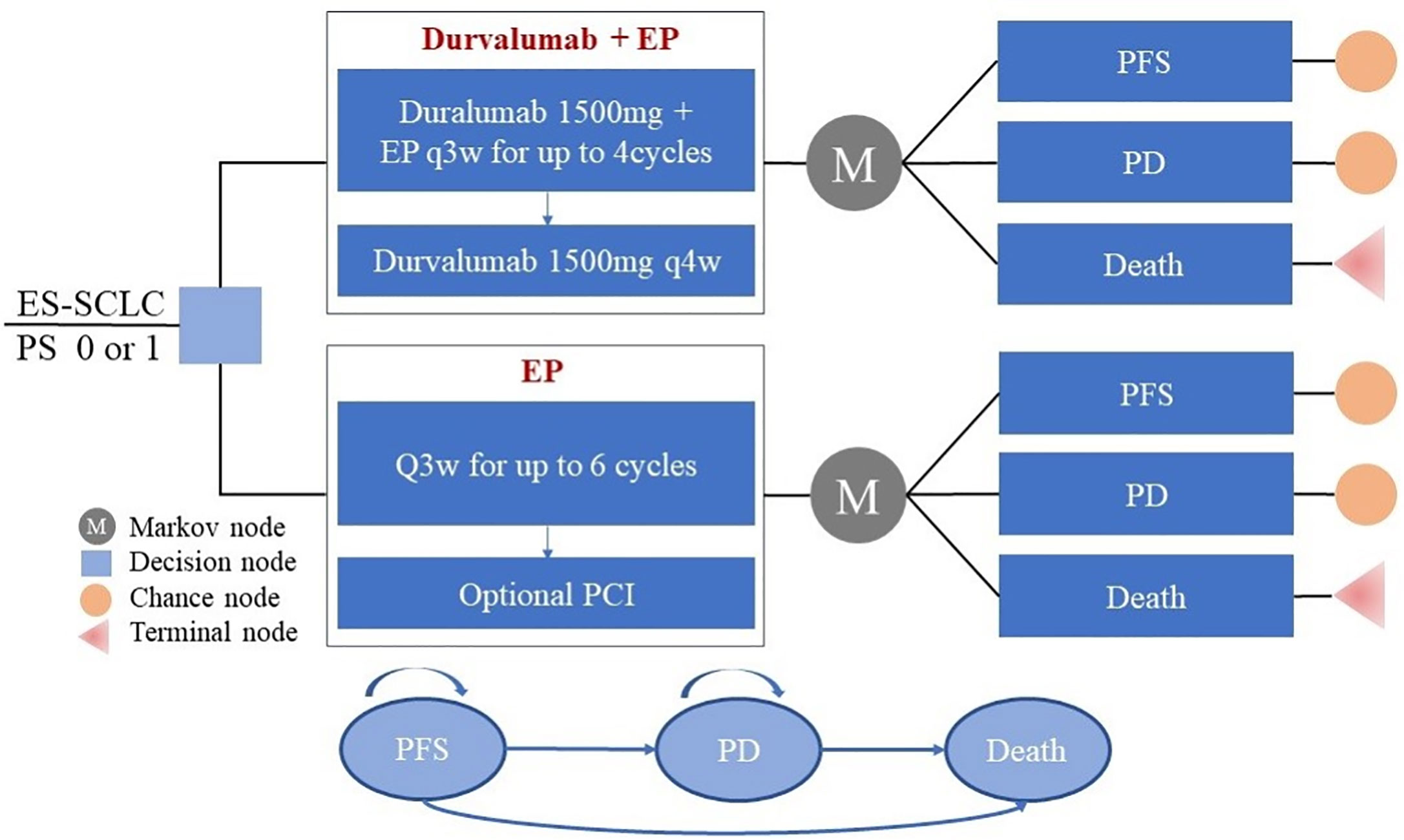
Frontiers | First-Line Durvalumab Plus Platinum-Etoposide Versus Platinum-Etoposide for Extensive-Stage Small-Cell Lung Cancer: A Cost-Effectiveness Analysis

Olaparib + durvalumab + tremelimumab combination therapy dosing schedule | Download Scientific Diagram

Durvalumab in NSCLC: latest evidence and clinical potential - Nerea Muñoz-Unceta, Isabel Burgueño, Elizabeth Jiménez, Luis Paz-Ares, 2018

Astrazenica Imfinzi Durvalumab Anti Cancer Injection, Dosage Form: 500MG at Rs 160000/vial in Gautam Budh Nagar

Durvalumab and Tremelimumab for Hepatocellular Carcinoma in Patients Listed for a Liver Transplant | SpringerLink

IMFINZI® (durvalumab) Plus Chemotherapy Approved in the US as the First Immunotherapy Regimen for Patients with Advanced Biliary Tract Cancer

Safety and antitumour activity of durvalumab plus tremelimumab in non-small cell lung cancer: a multicentre, phase 1b study - The Lancet Oncology

Primary results of STRONG: An open-label, multicenter, phase 3b study of fixed-dose durvalumab monotherapy in previously treated patients with urinary tract carcinoma - ScienceDirect

Cancers | Free Full-Text | Durvalumab after Sequential High Dose Chemoradiotherapy versus Standard of Care (SoC) for Stage III NSCLC: A Bi-Centric Retrospective Comparison Focusing on Pulmonary Toxicity
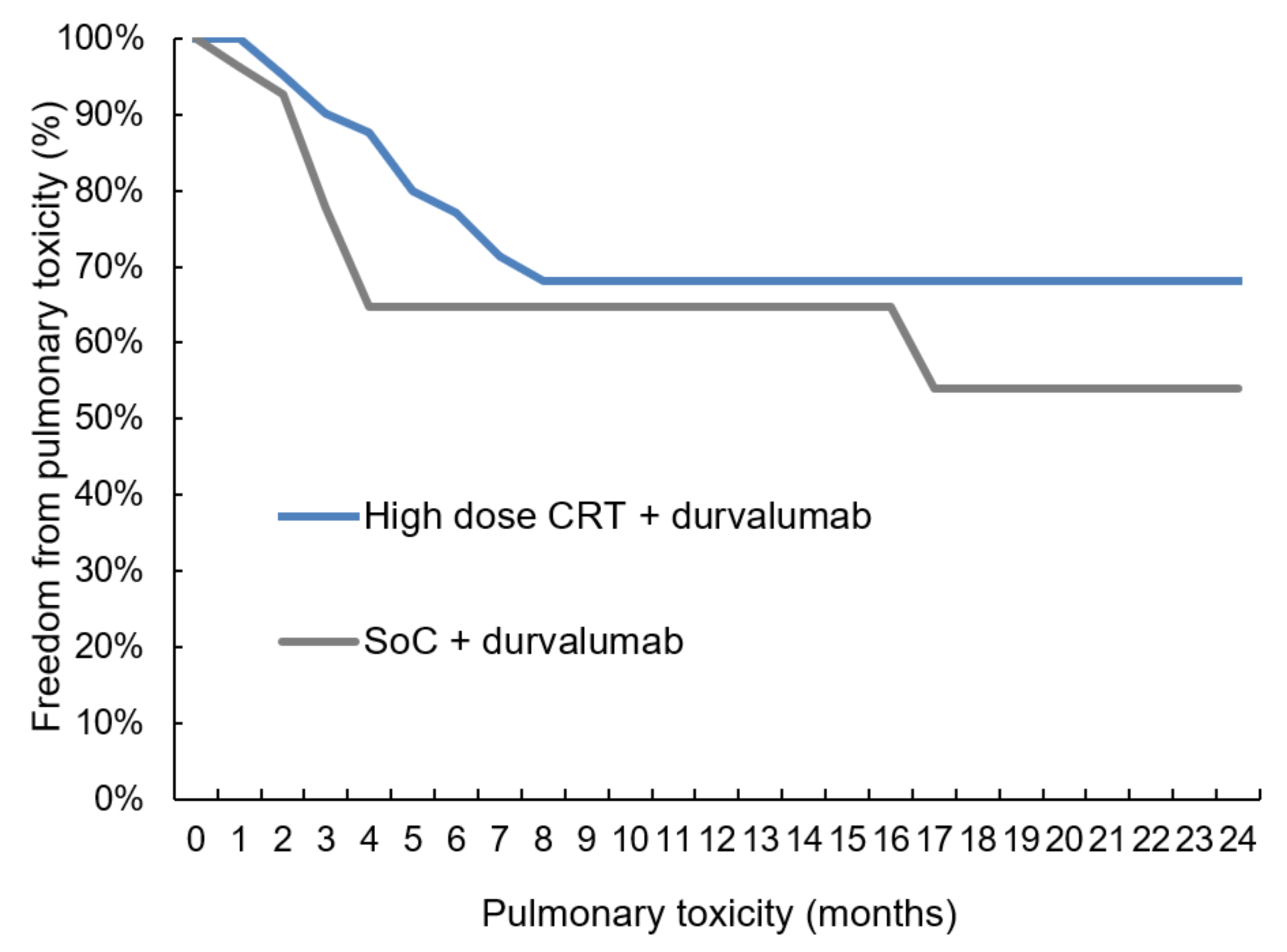
Cancers | Free Full-Text | Durvalumab after Sequential High Dose Chemoradiotherapy versus Standard of Care (SoC) for Stage III NSCLC: A Bi-Centric Retrospective Comparison Focusing on Pulmonary Toxicity

Astrazeneca Imfinzi (Durvalumab), Dosage Form: Injection, Single Use Vial at Rs 48000/vial in Patiala