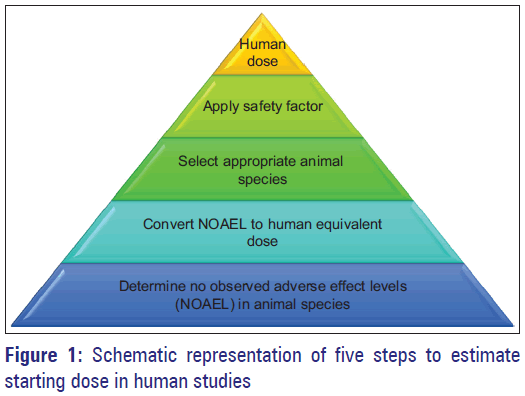
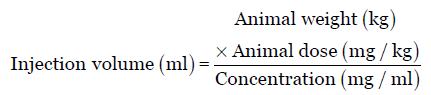![PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8750b5d4c6564d44ac84826e3760f15ea0b836de/22-Table3-1.png)
PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar
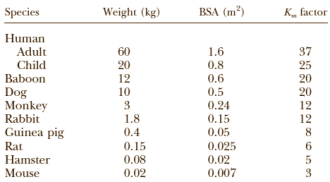
![PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8750b5d4c6564d44ac84826e3760f15ea0b836de/10-Table1-1.png)
PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar
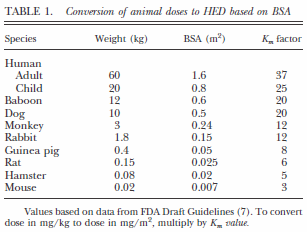
![PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f761b1fb3050f90ba97b77467f776594c3773982/5-Figure2-1.png)
PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar

Dose translation between laboratory animals and human in preclinical and clinical phases of drug development - Nair - 2018 - Drug Development Research - Wiley Online Library

Characterizing the human equivalent dose of herbal medicines in animal toxicity studies - ScienceDirect

Ask Dr. Andro: What Are Human Equivalent Doses (HED) and How Do I Calculate Them? - SuppVersity: Nutrition and Exercise Science for Everyone
![PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f761b1fb3050f90ba97b77467f776594c3773982/4-Figure1-1.png)